Respiratory Protection vs. Source Control – What’s the difference?
Posted on byIn an effort to limit the spread of COVID-19, CDC recommends that adults and children two years and older wear a mask, especially when social distancing is not possible or feasible[1]. Cloth masks and medical masks used in healthcare settings (such as surgical or procedure masks) are important tools in the fight against the spread of COVID-19, however they are not a type of respiratory protection. Due to limited supplies, respiratory protection should be reserved for workers who rely on it to perform their work safely. The masks that we all should wear are primarily intended for source control. What is the difference between respiratory protection and source control?
Respiratory protection protects the wearer. Respiratory protection refers to respirators, which are protective devices that cover a person’s nose and mouth or the entire face or head to help reduce the wearer’s exposure from breathing in air that contains contaminants, such as small respiratory droplets from a person who has COVID-19. This type of protection can include filtering facepiece respirators (FFRs), like N95 respirators.
Source control protects others. Source control refers to the use of masks to cover a person’s mouth and nose and to help reduce the spread of large respiratory droplets to others when the person talks, sneezes, or coughs. This can help reduce the spread of SARS-CoV-2, the virus that causes COVID-19, by someone who is infected but does not know it.
Breaking Down the Difference

Respiratory Protection: Respirators, such as the well-known N95 filtering facepiece respirator, remove particles from the inhaled airstream of the wearer through a filter media. When used in accordance with the manufacturer’s recommendations, respirators are highly effective at reducing the wearer’s exposure to bacteria and viruses, as well as other airborne particles like mold and dust, in their inhaled airstream. N95 filtering facepiece respirators are disposable devices that in times of normal operation, are discarded after every use or shift. There are key criteria for these personal protective devices to be effective as respiratory protection:
- The respirator filter needs to be highly effective at capturing particles that pass through the filter;
- The respirator must also fit the user’s face snugly to minimize the number of particles that bypass the filter and get into the breathing zone through gaps between the user’s skin and the respirator seal. A good seal helps ensure that the air goes through the filter.
When worn properly, respirators are designed to help protect the wearer by passing most of the air through the filter. N95 respirators filter at least 95% of particles of all sizes from inhaled air. The National Institute for Occupational Safety and Health (NIOSH) regulates respirators by using stringent test conditions to evaluate these devices, approving those that meet a minimum filtration efficiency requirement for occupational use.
It is important to note that a respirator that has earned NIOSH approval will have specific labeling printed on the facepiece. You may see some masks on the market labelled as “N95”, but if it does not contain the other components of the required label, most importantly the word “NIOSH,” it may not be NIOSH-approved and, therefore, cannot be relied on to provide respiratory protection. You can further ensure that you are using a NIOSH-approved respirator by checking for the model on the NIOSH Certified Equipment List or the NIOSH List of Approved Filtering Facepiece Respirators.
Additionally, if the respirator does not form a tight seal with the face, it cannot provide the expected level of protection. The Occupational Safety and Health Administration (OSHA) requires that workers are fit tested on the make and model of respirator that will be used, the first time it is used and every year after (or even sooner if the user has had a significant change in weight or change to their facial features, such as what might occur with extensive dental work). Fit testing ensures that respirator users are receiving the expected level of protection by minimizing contaminant leakage into the facepiece.
NOTE:
Preliminary data suggests that the outward leakage from exhalation valves is less than or comparable to that of many devices being used for source control (e.g., cloth masks, bandanas, surgical masks). However, until more research is available, masks with exhalation valves or vents should NOT be worn to help prevent the person wearing the mask from spreading COVID-19 to others (source control). Here are some tips when it comes to exhalation valves:
- Wear a respirator without an exhalation valve when both source control and respiratory protection are required.
- If only a respirator with an exhalation valve is available and source control is needed, cover the exhalation valve with a surgical mask or a cloth mask that does not interfere with the respirator fit.
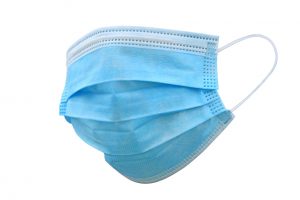
Source Control: Unlike respirators, masks are not designed to reduce the particles that the wearer will inhale and are not evaluated by NIOSH for their effectiveness to protect the wearer from airborne hazards. While there are many different mask designs available, they typically do not form the necessary seal against the wearer’s skin or have the appropriate level of filtration. Additionally, a typical mask wearer, outside of an occupational respiratory protection program, does not undergo fit testing to ensure an adequate level of protection. Therefore, while filtering facepiece respirators can be used as a type of source control (except those that have an exhalation valve in certain occupational settings), a mask should not be used as a respirator.
The purpose of wearing masks is to help reduce the spread of COVID-19 by reducing the spread of the virus through respiratory droplets from asymptomatic individuals. Masks are recommended as a barrier to help prevent large respiratory droplets from traveling into the air and onto other people when the person wearing the mask coughs, sneezes, talks, or raises their voice. Emerging evidence from clinical and laboratory studies shows that masks help reduce the spray of droplets when worn over the nose and mouth. Together with social distancing, masks are most likely to reduce the spread of COVID-19 when they are widely used by people in public settings.
While masks help to keep some large respiratory droplets contained and from reaching other people, they are not perfect. If you are sick with COVID-19 or think you might have COVID-19, wearing a mask does not make it safe or acceptable to visit public areas or be close to other people. Isolate yourself, whether or not you have symptoms. Stay home except when directed by your medical provider to get medical care. As much as possible, stay in a specific room and away from other people and pets in your home, use a separate bathroom if available, and do not share personal items like cups, towels or utensils. If you need to be around other people or animals within your own home, you should wear a mask, and be sure to frequently wash your hands. Those with underlying medical conditions who find it more difficult to breathe when wearing a mask can check with their health care provider to determine if a mask is appropriate for you. Masks should not be worn by children under the age of two or anyone who has trouble breathing, is unconscious, incapacitated, or otherwise unable to remove the mask without assistance.
A benefit of masks that are made from cloth material is that they can, and should, be washed after every use.
Should I Wear a Respirator?
The CDC does not recommend that the general public wear respirators to protect themselves from COVID-19. Due to the way SARS-CoV-2 spreads, the work duties of healthcare workers (e.g., directly caring for persons with suspected or known COVID-19), and possible supply shortages of respiratory protection, it is critical that those workers who need respiratory protection have it readily available to perform their work safely.
Additionally, the general public typically will not have undergone a fit test, therefore may not receive the expected level of protection from the respirator. Those required to wear respirators in the workplace must pass a medical evaluation to determine their fitness for wearing a respirator. However, a member of the general public may not have had such an evaluation, which may lead to complications for some people (e.g., those with underlying respiratory conditions) if they decide to wear a respirator.
A Note on Surgical Masks Used in Healthcare Settings
A surgical mask is a loose-fitting device that creates a physical barrier between the mouth and nose of the wearer and potential contaminants in the immediate environment. Surgical masks are regulated by the Food and Drug Administration (FDA) and are evaluated for their ability to protect the wearer from contact with liquids. Surgical masks are used as standard pieces of personal protective equipment in healthcare settings, including for COVID-19.
If worn properly, a surgical mask is meant to help block large-particle droplets, splashes, sprays, or splatter that may contain germs (viruses and bacteria), keeping it from reaching the wearer’s mouth and nose. Surgical masks also act as source control, just as any other mask. While they are evaluated by the FDA for their ability to protect against contact with liquids, surgical masks are not designed to filter or block very small particles from the air that the wearer is breathing. Unlike respirators, they do not provide complete protection because they do not form a tight seal against the wearer’s face.
Important Reminder
The best way to prevent COVID-19 illness is to avoid being exposed to this virus. In addition to wearing a mask, the CDC recommends everyday preventive actions, including staying home if you have been exposed or are sick, staying at least 6 feet apart from others, and hand hygiene in order to help prevent the spread of respiratory diseases.
Jaclyn Krah Cichowicz, MA, is a Health Communications Specialist in the in the NIOSH National Personal Protective Technology Laboratory.
LCDR Megan Casey, RN, BSN, MPH, is a Nurse Epidemiologist in the NIOSH National Personal Protective Technology Laboratory.
Maryann M. D’Alessandro, PhD, is the Director of the NIOSH National Personal Protective Technology Laboratory.
[1] Masks should NOT be worn by children under the age of 2 or anyone who has trouble breathing, is unconscious, incapacitated, or otherwise unable to remove the mask without assistance.
Posted on by

