Category: pharmacogenomics
Cost-effectiveness of Pharmacogenomic Testing: How to Measure the Value of Having the Right Dose of the Right Drug for the Right Patient

A recent systematic review that assessed the cost-effectiveness of pharmacogenetic testing for drugs with existing guidelines concluded that most studies favored pharmacogenomic testing. The significance of this conclusion must be interpreted with caution and in the context of study factors, such as funding sources, geography, cohort, and the cost-effectiveness comparisons being made. Pharmacogenomics (PGx) combines Read More >
Posted on byUsing Pharmacogenomics to Better Understand the Role of Selected Medications and Birth Defect Risk

Through a funding opportunity from CDC’s Office of Genomics and Precision Public Health in collaboration with the Office of Advanced Molecular Detection, CDC’s Birth Defects Monitoring and Research Branch in the National Center on Birth Defects and Developmental Disabilities will conduct a 2-year project to gather genome-wide genotyping data to look at relationships between pharmacogenomic Read More >
Posted on byTracking the Scientific Literature on the Impact of Pharmacogenomics on Clinical Practice and Public Health
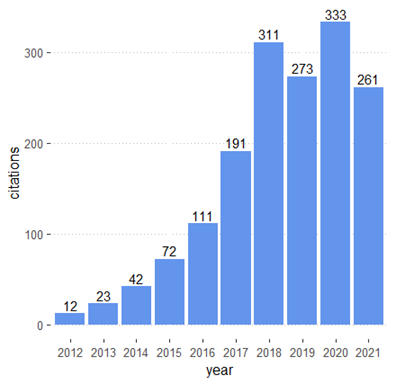
Pharmacogenomics (PGx) is an emerging field that investigates genetic differences in drug effectiveness and safety. PGx is an essential component of precision medicine and seeks to provide the right medication for the right person at the right time. Advances in PGx promise to improve treatment of many diseases such as cancer and cardiovascular disease. In Read More >
Posted on byAssessing Pharmacogenetic Variation in the United States to Enhance Health Equity of Pharmacogenetic Testing
CDC’s Genetic Testing Reference Material program (GeT-RM), in the Division of Laboratory Systems will conduct a 2-year project to test 5000 DNA samples from the population-based National Health and Nutrition Examination Survey (NHANES) to determine the allele frequencies of 970 unique haplotypes in 150 pharmacogenes selected because they have high clinical annotation levels of evidence Read More >
Posted on byUsing Pharmacogenetics to Enhance Tuberculosis (TB) Treatment

Through a funding opportunity from CDC’s Office of Genomics and Precision Public Health in collaboration with the CDC Office of Advanced Molecular Detection, CDC’s Division of Tuberculosis Elimination will conduct a 2-year project to assess relationships between pharmacogenetics (PG), TB drug exposure, relevant treatment outcomes, and safety. Researchers will use information collected in an international phase Read More >
Posted on byIntegrating Pharmacogenomics in Practice: One GIFT at a Time or a Package Deal?

Testing for genetic variants related to warfarin metabolism has been the poster child for pharmacogenomics in clinical practice for over a decade. A recent publication of the Genetic Informatics Trial (GIFT) provides additional evidence for the clinical benefits of incorporating pharmacogenomics testing into the management of warfarin initiation. In this multicenter study of older individuals Read More >
Posted on byEvidence Matters in Genomic Medicine- Round 4: Where are we with Pharmacogenomic Tests?

Previously, CDC’s Office of Public Health Genomics announced a list of health-related genomic tests and applications, stratified into three tiers according to the availability of scientific evidence and evidence-based recommendations and systematic reviews. The list is intended to promote information exchange and dialogue among researchers, providers, policy makers, and the public. Initially the table relied Read More >
Posted on byMedications for the Masses? Pharmacogenomics is an Important Public Health Issue

Adverse drug events are a serious public health problem. Consider the following facts: an estimated 82% of American adults take at least one medication and 29% take five or more; 700,000 emergency department visits and 120,000 hospitalizations are due to adverse drug events annually; $3.5 billion is spent on extra medical costs of adverse drug Read More >
Posted on by 3 Comments

