Category: genomics
Get up to Speed on the Latest Developments in the Field! Register for the ORISE Current Issues in Genomics and Precision Public Health Online Training Event, September 7–8, 2023.

Advances in genomics, data science, machine learning, and artificial intelligence are transforming practice. Next generation public health and medical workforces need to understand these developments and how they can be used to benefit population health. Recognizing this challenge, Oak Ridge Institute for Science and Education (ORISE) is partnering with the Office of Genomics and Precision Read More >
Posted on byPersistent Underutilization of BRCA Testing for Breast and Ovarian Cancer in the United States: Implications for Health Disparities

Two recent studies document ongoing underutilization and disparities in genetic testing for patients with breast and ovarian cancer. BRCA1 and BRCA2 (BRCA) genetic testing is supported by clinical guidelines to inform hereditary cancer risk for people with certain personal or family health histories of breast, ovarian, pancreatic, or prostate cancer, and to guide treatment decisions Read More >
Posted on by 1 CommentThe Promise of Population-based Genomic Screening for Selected Hereditary Conditions: Contributions of Cost-Effectiveness Analysis

It is estimated that 3 million people in the United States carry pathogenic variants that increase their risks for heart disease and cancer. If people with such variants are identified, medical interventions are available to significantly reduce morbidity and mortality. However, existing recommendations tend to emphasize family-based or ethnic-specific criteria to determine at-risk individuals for Read More >
Posted on by 1 CommentData for Action in Public Health Genomics: Ensuring Equitable Implementation of Genomic Applications Across the Lifespan

In the more than 20 years since the completion of the Human Genome Project, basic and clinical research have delivered on the promise to develop genomic applications that can help prevent and treat many diseases across the lifespan. However, efforts to ensure equitable implementation of genomic applications have fallen short particularly among racial and ethnic Read More >
Posted on byPopulation-based Genomic Screening Programs: The Need for Optimal Implementation to Ensure Health Equity

Population genomic screening of adults has emerged as a strategy to promote prevention of common diseases such as cancer and heart disease among persons with genetic conditions. This type of screening has the potential to identify millions of currently undetected people in the United States who are at risk of preventable diseases for which evidence-based Read More >
Posted on byTracking the Contributions of Implementation Science to the Population Health Impact of Genomics and Precision Health: A New Knowledge Base
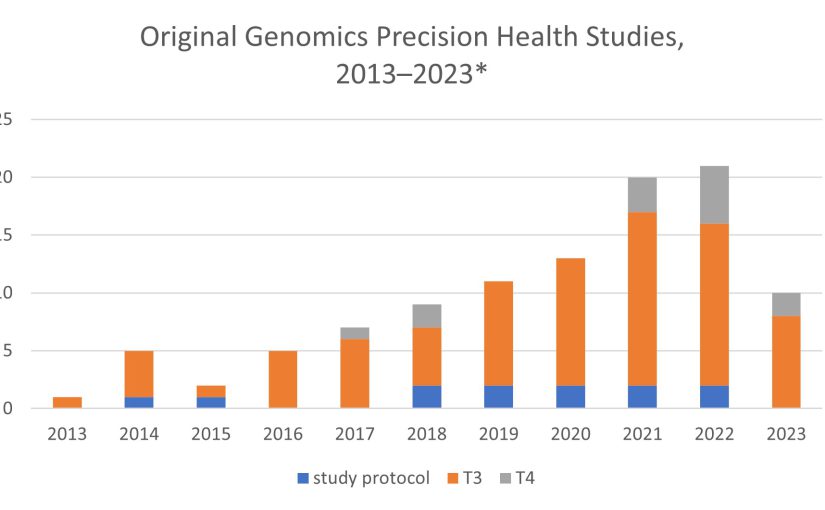
Successful implementation of evidence-based genomic and precision health interventions requires an understanding of what works and what doesn’t work within the context of various clinical and public health settings. Research and evaluation that incorporate implementation science tools and methods into the translation of these interventions facilitate this. Here we present a new CDC knowledge base, Read More >
Posted on by 1 CommentCost-effectiveness of Pharmacogenomic Testing: How to Measure the Value of Having the Right Dose of the Right Drug for the Right Patient

A recent systematic review that assessed the cost-effectiveness of pharmacogenetic testing for drugs with existing guidelines concluded that most studies favored pharmacogenomic testing. The significance of this conclusion must be interpreted with caution and in the context of study factors, such as funding sources, geography, cohort, and the cost-effectiveness comparisons being made. Pharmacogenomics (PGx) combines Read More >
Posted on byRare Primary Immunodeficiency Diseases and COVID-19: Evolving Insights and Implications for Clinical and Public Health Practice

In this post, we explore how new research on rare genetic diseases is contributing to our understanding of COVID-19 occurrence and outcomes and discuss potential clinical and public health implications. Understanding the mechanisms involved in these inherited disorders may shed light on biological mechanisms and natural history of COVID-19. Primary Immunodeficiency and COVID-19 Primary immunodeficiency (PI)—often Read More >
Posted on byInterplay Between the Exposome and the Genome in Health and Disease
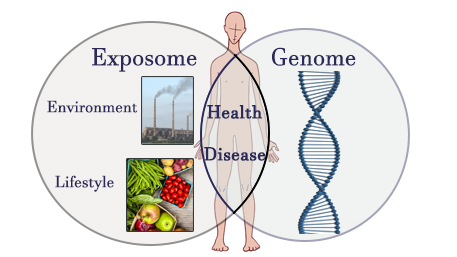
A recent review assessed the interplay between environmental exposures and the human genome and showed ways that this interplay can alter disease risk. Many diseases, such as birth defects and developmental disabilities, type 2 diabetes and cancer, are influenced by both environmental and genetic factors. The cumulative effects of environmental exposures prenatally and throughout life Read More >
Posted on byPopulation Screening for Rare Pathogenic Variants as the Low Hanging Fruit for Public Health Genomics Across the Lifespan

Rapid advances in human genome sequencing technologies have accelerated the integration of genomics into clinical practice. Genomics has demonstrated clinical utility as a diagnostic tool for certain diseases, but its potential for population screening is still work in progress. In principle, DNA-based population screening can identify persons with rare pathogenic variants who are at high Read More >
Posted on by 1 Comment

