Epigenetics and Public Health: Why We Should Pay Attention
Posted on byIn September 2014, one of us (MJK) spoke on the topic of epigenetics at the Annual Meeting of the Association for State and Territorial Health Officials (ASTHO). ASTHO is a national organization representing public health agencies in the United States. ASTHO members formulate and influence public health policy and practice. In the midst of a busy agenda, state health officials were interested in learning about epigenetics as a new and evolving area for public health practice. Why is that?
At the CDC Office of Public Health Genomics, we have been tracking the progress of genomics and related fields and their impact on clinical practice and disease prevention. In our evidence-based table and recent highlights of the year, we have mentioned an increasing number of genomic tests and applications for which evidence-based recommendations exist and can save lives today. Epigenetics so far has not made it on the list!
So what is epigenetics and why is it attracting interest these days? This is obviously a “hot” topic in research and the media. Here are a few recent headlines
Can you inherit experiences? Inside the weird world of epigenetics
You are what you eat, but what about your DNA?
Sperm contains dad’s lifestyle information alongside basic genetic material
Epigenetics: genes, environment and the generation game: “New research claims that environmental factors affect not just an individual’s genes but those of their offspring too. Diabetes, obesity – even certain phobias – may all be influenced by the behavior of our forebears.”
It turns out we all have two “biological codes” that are important in development, health and disease. The genetic code is the sequence of DNA (base pairs) that tells a cell how to build proteins, the essential building blocks of life. More than 99.9% of the genetic code is identical among us, and the 0.1% variation is important in health traits such as height, weight and eye color. Genetic variation is also important in individual susceptibility to various diseases across the life span, including the thousands of rare “genetic diseases” such as sickle cell disease and cystic fibrosis in which single mutations (genetic changes) can have drastic effects on health and disease.
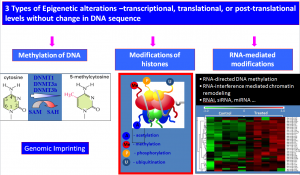
The epigenetic code (literally means “above” the genome) does not affect the information contained in DNA sequence, but controls when and where this information is available to cells. The epigenetic code is determined by several mechanisms that affect gene expression, the most well-known of which is DNA methylation (See Figure 1). The epigenetic code can be altered by environmental exposures such as chemicals, nutrition and stress, especially in early life. Such exposures can have a profound and long lasting impact on gene expression across generations.
Most of our knowledge about epigenetics comes from experimental studies. In humans, epigenetic changes in cancer are among the best characterized. Epigenetics can also be involved in certain birth defects that can be affected by nutritional factors, such as deficiency of folic acid in the diet.

To illustrate the dramatic impact of epigenetics and the influence of modifiable environmental factors, let us look briefly at the example of the Agouti mouse. Figure 2 shows two mice with identical genetic codes (as with identical twins). In the yellow mouse, a region of the DNA is unmethylated which makes the nearby Agouti gene turned on all the time, while in the brown mouse, the region is methylated and the Agouti gene is turned off. The turning on of this gene leads not only to changes in coat color but to predisposition to all kinds of metabolic diseases including obesity and diabetes. Environmental exposures can influence whether or not this gene is turned on or off. For example, when pregnant mice are fed bisphenol A (BPA), a chemical present in many commonly used products, the number of offspring with the yellow obese coat color increases dramatically. This happens because BPA decreases DNA methylation so that more offspring have unmethylated Agouti genes. Thus, BPA exposure is associated with a higher number of yellow mice predisposed to obesity and diabetes. When pregnant mice are exposed to BPA along with a vitamin B cocktail (including B12 and folic acid) which increase methylation of the Agouti gene, the offspring are no longer predominantly yellow and obese. Folic acid is important in growth and development and is recommended for women in the prevention of selected birth defects (notably neural tube defects such as spina bifida). Humans have many regions of the genome that are susceptible to epigenetic regulation.
There is also strong suspicion that cumulative epigenetic changes due to environmental exposures and stressors can help explain health disparities in the burden of various diseases among disadvantaged populations. Measuring neighborhood-specific epigenetic alterations potentially can be used to investigate the mechanisms underlying health disparities.
Can we use epigenetic information to understand and prevent human disease through environmental modifications (reducing exposures, dietary changes and medication use)? The utility of epigenetic information in improving health should follow evidence-based approaches such as the one laid out by the CDC ACCE framework and developed further by the EGAPP working group for using genomic tests. Broad areas of inquiry include 1) assessing accuracy of measurement of epigenetic alterations and their transmission (analytic validity), 2) assessing the relationship between epigenetic alterations to environmental exposures as well as health outcomes (clinical validity); and 3) evaluating the use of epigenetic biomarkers in environmental risk assessment and interventions.
To succeed in identifying epigenetic mechanisms that can lead to human diseases, public health researchers must integrate animal models with clinical and epidemiologic studies, focusing on accuracy of measurements, cell and organ-specific epigenetic patterns as well as windows of vulnerability for epigenetic phenomena in relation to environmental exposures in large prospective studies.
The potential of impact of epigenetic knowledge will be specific to each health outcome. Effective prevention and treatment await a more complete understanding of the causes of human disease and the role that epigenetic modifications can play in improving the health of individuals and populations.
Posted on by


